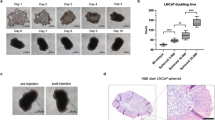
Abstract
LuCaP serially transplantable patient-derived xenografts (PDXs) are valuable preclinical models of locally advanced or metastatic prostate cancer. Using spheroid culture methodology, we recently established cell lines from several LuCaP PDXs. Here, we characterized in depth the features of xenografts derived from LuCaP 136 spheroid cultures and found faithful retention of the phenotype of the original PDX. In vitro culture enabled luciferase transfection into LuCaP 136 spheroids, facilitating in vivo imaging. We showed that LuCaP 136 spheroids formed intratibial, orthotopic, and subcutaneous tumors when re-introduced into mice. Intratibial tumors responded to castration and were highly osteosclerotic. LuCaP 136 is a realistic in vitro–in vivo preclinical model of a subtype of bone metastatic prostate cancer.









Similar content being viewed by others
Abbreviations
- AR:
-
Androgen receptor
- H&E:
-
Hematoxylin and eosin
- HBS:
-
HEPES-buffered saline
- IHC:
-
Immunohistochemistry
- K5:
-
Keratin 5
- K18:
-
Keratin 18
- MRI:
-
Magnetic resonance imaging
- PCa:
-
Prostate cancer
- PDX:
-
Patient-derived xenograft
- PINP:
-
N-terminal propeptide of procollagen type 1a
- PSA:
-
Prostate-specific antigen
- Te:
-
Testosterone
- μCT:
-
Micro-computed tomography
References
Weilbaecher KN, Guise TA, McCauley LK (2011) Cancer to bone: a fatal attraction. Nat Rev Cancer 11(6):411–425
Groot MT et al (2003) Costs of prostate cancer, metastatic to the bone, in the Netherlands. Eur Urol 43(3):226–232
Lange PH, Vessella RL (1998) Mechanisms, hypotheses and questions regarding prostate cancer micrometastases to bone. Cancer Metastasis Rev 17(4):331–336
Parker C et al (2013) Alpha emitter radium-223 and survival in metastatic prostate cancer. N Engl J Med 369(3):213–223
Mohler JL et al (2014) Prostate cancer, version 2.2014. J Natl Compr Canc Netw 12(5):686–718
Crawford ED et al (2015) Treating patients with metastatic castration resistant prostate cancer: a comprehensive review of available therapies. J Urol 194(6):1537–1547
Ocana A et al (2011) Preclinical development of molecular-targeted agents for cancer. Nat Rev Clin Oncol 8(4):200–209
Sausville EA, Burger AM (2006) Contributions of human tumor xenografts to anticancer drug development. Cancer Res 66(7):3351–3354 (discussion 4)
Sobel RE, Sadar MD (2005) Cell lines used in prostate cancer research: a compendium of old and new lines–part 1. J Urol 173(2):342–359
Najy AJ et al (2012) Cediranib inhibits both the intraosseous growth of PDGF D-positive prostate cancer cells and the associated bone reaction. Prostate 72(12):1328–1338
Valta MP et al (2008) FGF-8 is involved in bone metastasis of prostate cancer. Int J Cancer 123(1):22–31
Bonfil RD et al (2007) Prostate cancer-associated membrane type 1-matrix metalloproteinase: a pivotal role in bone response and intraosseous tumor growth. Am J Pathol 170(6):2100–2111
van Weerden WM, Bangma C, de Wit R (2009) Human xenograft models as useful tools to assess the potential of novel therapeutics in prostate cancer. Br J Cancer 100(1):13–18
Young SR et al (2013) Establishment and serial passage of cell cultures derived from LuCaP xenografts. Prostate 73(12):1251–1262
Barbieri CE et al (2012) Exome sequencing identifies recurrent SPOP, FOXA1 and MED12 mutations in prostate cancer. Nat Genet 44(6):685–689
Saar M et al (2014) Spheroid culture of LuCaP 147 as an authentic preclinical model of prostate cancer subtype with SPOP mutation and hypermutator phenotype. Cancer Lett 351(2):272–280
Ibrahim L, Wright EA (1983) Effect of castration and testosterone propionate on mouse vibrissae. Br J Dermatol 108(3):321–326
Saar M et al (2012) Experimental orthotopic prostate tumor in nude mice: techniques for local cell inoculation and three-dimensional ultrasound monitoring. Urol Oncol 30(3):330–338
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH Image to ImageJ: 25 years of image analysis. Nat Methods 9(7):671–675
Kumar A et al (2011) Exome sequencing identifies a spectrum of mutation frequencies in advanced and lethal prostate cancers. Proc Natl Acad Sci USA 108(41):17087–17092
Wettenhall JM, Smyth GK (2004) limmaGUI: a graphical user interface for linear modeling of microarray data. Bioinformatics 20(18):3705–3706
Leek JT et al (2012) The sva package for removing batch effects and other unwanted variation in high-throughput experiments. Bioinformatics 28(6):882–883
Leeming DJ et al (2011) Serum N-terminal propeptide of collagen type i is associated with the number of bone metastases in breast and prostate cancer and correlates to other bone related markers. Biomark Cancer 3:15–23
Edwards JR, Mundy GR (2011) Advances in osteoclast biology: old findings and new insights from mouse models. Nat Rev Rheumatol 7(4):235–243
Zhang X et al (2015) Amplification and protein expression of androgen receptor gene in prostate cancer cells: fluorescence hybridization analysis. Oncol Lett 9(6):2617–2622
Ayala G et al (2015) Expression of ERG protein in prostate cancer: variability and biological correlates. Endocr Relat Cancer 22(3):277–287
Adley BP, Yang XJ (2006) Application of alpha-methylacyl coenzyme A racemase immunohistochemistry in the diagnosis of prostate cancer: a review. Anal Quant Cytol Histol 28(1):1–13
Simmons JK et al (2015) Animal models of bone metastasis. Vet Pathol 52(5):827–841
Ganguly SS, Li X, Miranti CK (2014) The host microenvironment influences prostate cancer invasion, systemic spread, bone colonization, and osteoblastic metastasis. Front Oncol 4:364
Nemeth JA et al (1999) Severe combined immunodeficient-hu model of human prostate cancer metastasis to human bone. Cancer Res 59(8):1987–1993
Graham TJ et al (2014) Preclinical evaluation of imaging biomarkers for prostate cancer bone metastasis and response to cabozantinib. J Natl Cancer Inst 106(4):dju033
Yang J et al (2001) Prostate cancer cells induce osteoblast differentiation through a Cbfa1-dependent pathway. Cancer Res 61(14):5652–5659
Hagberg Thulin M et al (2014) Osteoblasts stimulate the osteogenic and metastatic progression of castration-resistant prostate cancer in a novel model for in vitro and in vivo studies. Clin Exp Metastasis 31(3):269–283
Li ZG et al (2008) Androgen receptor-negative human prostate cancer cells induce osteogenesis in mice through FGF9-mediated mechanisms. J Clin Investig 118(8):2697–2710
Roychowdhury S et al (2011) Personalized oncology through integrative high-throughput sequencing: a pilot study. Sci Transl Med 3(111):111ra21
Larson SR et al (2013) Characterization of osteoblastic and osteolytic proteins in prostate cancer bone metastases. Prostate 73(9):932–940
Brennecke P et al (2014) CXCR4 antibody treatment suppresses metastatic spread to the lung of intratibial human osteosarcoma xenografts in mice. Clin Exp Metastasis 31(3):339–349
Drake CG (2014) Visceral metastases and prostate cancer treatment: ‘die hard’, ‘tough neighborhoods’, or ‘evil humors’? Oncology (Williston Park) 28(11):974–980
Akfirat C et al (2013) Tumour cell survival mechanisms in lethal metastatic prostate cancer differ between bone and soft tissue metastases. J Pathol 230(3):291–297
Morrissey C et al (2010) Inhibition of angiopoietin-2 in LuCaP 23.1 prostate cancer tumors decreases tumor growth and viability. Prostate 70(16):1799–1808
Brubaker KD et al (2006) Administration of zoledronic acid enhances the effects of docetaxel on growth of prostate cancer in the bone environment. BMC Cancer 6:15
Michiel Sedelaar JP, Dalrymple SS, Isaacs JT (2013) Of mice and men–warning: intact versus castrated adult male mice as xenograft hosts are equivalent to hypogonadal versus abiraterone treated aging human males, respectively. Prostate 73(12):1316–1325
Gonzalez-Billalabeitia E et al (2014) Vulnerabilities of PTEN-TP53-deficient prostate cancers to compound PARP-PI3K inhibition. Cancer Discov 4(8):896–904
Semenas J et al (2014) The role of PI3K/AKT-related PIP5K1alpha and the discovery of its selective inhibitor for treatment of advanced prostate cancer. Proc Natl Acad Sci USA 111(35):E3689–E3698
Acknowledgments
This work was supported by the Stanford Department of Urology [HZ, RN, DMP], funding for university level health research in Finland [MPV], a Ferdinand Eisenberger Grant of the German Society of Urology ID SaM1/FE-11 and HOMFOR (Saarland University, Germany) [MS], South-Western Cancer Foundation of Finland and K Albin Johansson Foundation, Finland [JT], Academy of Finland and Sigrid Jusélius Foundation, Finland [PH], The Richard Lucas Foundation and the PNW Prostate Cancer SPORE NIH P50 CA097186 [EC], NIH 2P50CA097186 [IC, PSN, EC], NIH 1R01CA165573 [IC, PSN, EC], NIH P01CA163227 [IC, PSN, EC], and the Prostate Cancer Foundation [EC, DMP]. Pharmatest Services LTD, Turku, Finland, is acknowledged for serum analyses and histological stainings.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No potential conflicts of interest were disclosed.
Rights and permissions
About this article
Cite this article
Valta, M.P., Zhao, H., Saar, M. et al. Spheroid culture of LuCaP 136 patient-derived xenograft enables versatile preclinical models of prostate cancer. Clin Exp Metastasis 33, 325–337 (2016). https://doi.org/10.1007/s10585-016-9781-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10585-016-9781-2